Conserved role of intragenic DNA methylation in regulating alternative promoters.
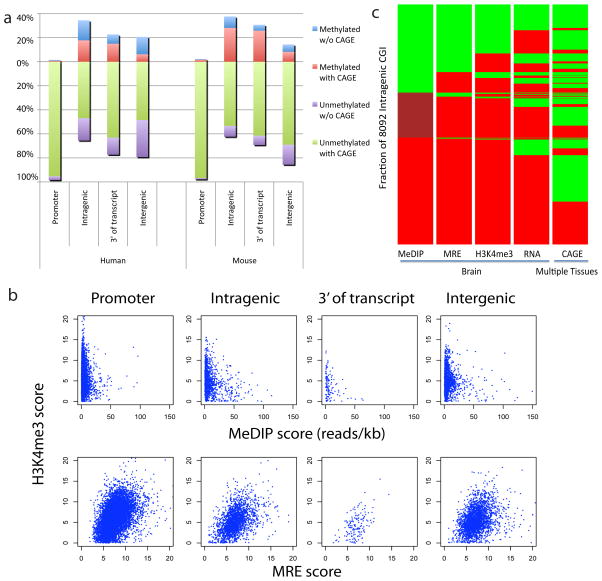
Although it’s identified that the methylation of DNA in 5′ promoters suppresses gene expression, the role of DNA methylation in gene our bodies is unclear. In mammals, tissue- and cell type-specific methylation is current in a small proportion of 5′ CpG island (CGI) promoters, whereas a far better proportion happens throughout gene our bodies, coinciding … Read more